Ph strips
10 posts
• Page 1 of 1
Ph strips
Is it important what kind of ph strips I buy? For example, does it matter if the strips are for urine and saliva?
An Englishman living in México.
There'e not such thing as a stupid question, just a stupid answer!
There'e not such thing as a stupid question, just a stupid answer!
- senorkevin
- Registered Member
- Posts: 354
- Joined: Tue May 01, 2012 12:47 pm
- Location: Mexico
Re: Ph strips
Doesn't matter as long as they have the right range.
Fashionably late will be stylishly hungry.
- NCPaul
- Site Admin
- Posts: 2922
- Joined: Thu Oct 01, 2009 12:58 am
- Location: North Carolina
Re: Ph strips
Paul, how would you prepare the salami sample if using urine strips?
Phil
Phil
-

wheels - Global Moderator
- Posts: 12891
- Joined: Sat Sep 02, 2006 4:29 pm
- Location: Leicestershire, UK
Re: Ph strips
I just tried it with the ones we use at work on a finished salami. I cut a fresh surface and pressed the plastic test strip against the meat; very poor transfer due to low moisture. I took the test strip and wetted it, then patted it dry and then used it; this gave a measurement of pH (as good as the test strips were able to resolve). I can see now the difficulty people have using test papers and I would not recommend them.
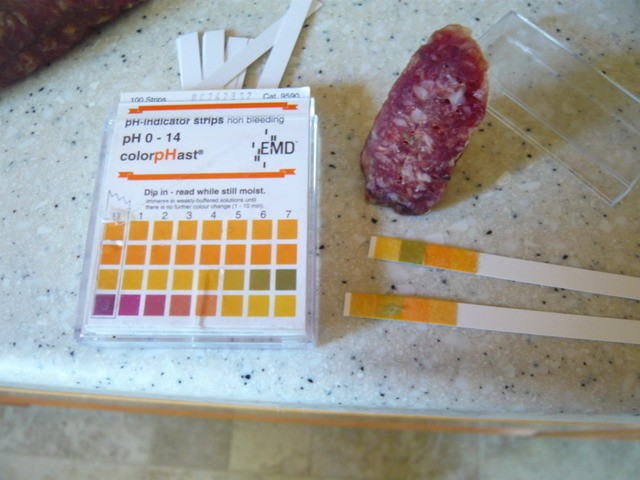

Fashionably late will be stylishly hungry.
- NCPaul
- Site Admin
- Posts: 2922
- Joined: Thu Oct 01, 2009 12:58 am
- Location: North Carolina
Re: Ph strips
The strips I have ( which I've yet to use ) recommends mixing a small sample of meat with some water, then test
http://www.butcher-packer.com/index.php ... cts_id=797
http://www.butcher-packer.com/index.php ... cts_id=797
-

DanMcG - Registered Member
- Posts: 1461
- Joined: Mon Dec 15, 2008 11:09 pm
- Location: Central NY, USA
Re: Ph strips
I'd appreciate Paul's opinion on that method Dan. Does it need any adjustment for the dilution, etc?
Phil
Phil
-

wheels - Global Moderator
- Posts: 12891
- Joined: Sat Sep 02, 2006 4:29 pm
- Location: Leicestershire, UK
Re: Ph strips
The first thing to remember about pH measurements is that they are on a -log10 scale. Changes in concentration require a correction. As an example, if I dilute my sample by half with deionized water and measure the pH as 5.0, the pH corrected for dilution would be 4.7. If I dilute my sample with three parts of deionized water to one part of meat and measure the pH as 5.0, the pH corrected for dilution would be 4.4. These are just approximations.  A better calculation would use the pKa of lactic acid and solve with a quadratic equation (hint, my way is simpler).
A better calculation would use the pKa of lactic acid and solve with a quadratic equation (hint, my way is simpler).
Fashionably late will be stylishly hungry.
- NCPaul
- Site Admin
- Posts: 2922
- Joined: Thu Oct 01, 2009 12:58 am
- Location: North Carolina
Re: Ph strips
Thanks Paul,
That set me thinking, so I had a delve around the net and found this calculator:
http://rechneronline.de/chemie-rechner/ph-dilution.php
I hope this helps Senorkevin
Phil
That set me thinking, so I had a delve around the net and found this calculator:
http://rechneronline.de/chemie-rechner/ph-dilution.php
I hope this helps Senorkevin
Phil
-

wheels - Global Moderator
- Posts: 12891
- Joined: Sat Sep 02, 2006 4:29 pm
- Location: Leicestershire, UK
Re: Ph strips
Cool Phil, Thanks for the link!
and thank you Paul for the education...........again.
and thank you Paul for the education...........again.
-

DanMcG - Registered Member
- Posts: 1461
- Joined: Mon Dec 15, 2008 11:09 pm
- Location: Central NY, USA
Re: Ph strips
That looks great. Thanks guys. The down side of me using the strips is that I'm colour blind 
I was thinking of getting strips that range from 3.9-5.7 and 4.9-6.9
If you wouldn't recommend the strips that what would you?
I was thinking of getting strips that range from 3.9-5.7 and 4.9-6.9
If you wouldn't recommend the strips that what would you?
An Englishman living in México.
There'e not such thing as a stupid question, just a stupid answer!
There'e not such thing as a stupid question, just a stupid answer!
- senorkevin
- Registered Member
- Posts: 354
- Joined: Tue May 01, 2012 12:47 pm
- Location: Mexico
10 posts
• Page 1 of 1
Who is online
Users browsing this forum: No registered users and 6 guests

